Soil Fertility
Introduction
Soil Fertility refers to the level of mineral nutrients essential for plant growth and development, and the chemical characteristics that affect nutrient availability to plants. Those nutrients listed in the table below are the ones that are most often limiting to plants in agricultural and to a lesser extent to forestry land management systems. Although managing soil fertility by the application of inorganic fertilizers and/or organic matter are common agricultural practices, the use of fertilizers and nutrient management practices are far less common in forestry and forest farming applications. None the less, understanding soil fertility-related limitations on your forest farming site is an important aspect of selecting appropriate non-timber forest crops.
Soil Nutrient Elements and Their Ionic (electrically changed: + or -) Forms That Are Available to Plants
| Soil nutrient element | Avaialable (ionic) form |
Soil pH and other chemical characteristics that affect nutrient ion availability (see Table below) |
| Nitrogen (N) |
NH4+ (ammonium)
NO3- (nitrate) |
NO3- readily leached; NH4+ retention increase on soils with high CEC (high organic matter, and/or high clay content) |
| Phosphorous (P) |
H2PO4- or
HPO4-2 (phosphate) |
Unavailable (fixed) at low pH, and in clayey soils |
| Potassium (K) |
K+ |
|
| Calcium (Ca) |
Ca+2 |
More abundant in high pH soils |
| Iron (Fe) |
Fe-2 (ferrous)
Fe-3 (ferric) |
Red soil color (uniform or mottling) indicates ferric (oxidized) Fe, and blue/green indicates ferrous (reduced) iron |
Nutrient Analysis (Soil Testing)
Although test kits for soil pH are available from garden centers, etc. complete soil chemical analysis is generally performed by a private, university or other soil analysis laboratory. Soil analysis usually includes levels of mineral nutrients (P, K, Ca, Mg, Zn, Fe, Al, Mn, and nitrate), pH and organic matter. Soil test kits can be obtained from the official testing laboratory in your state. It is important to follow instructions for collecting and submitting soil samples in order to obtain reliable results. Soil analysis reports include the results of the chemical analyses, often compared to a normal range for each nutrient. In some cases fertilizer recommendations are included if you have specified a particular crops with your application form. It is recommended that you should consult your local cooperative extension agent for complete interpretation of the results. Ultimately, because fertilizing forest soils is not often practiced, the most useful information you are likely to gain from soil analysis pH and OM content of your soil.
Soil pH
pH refers to the level of acidity or alkalinity of a soil based on the concentration of hydrogen ions (H+) in water, or
Chemically speaking, pH = -log [H+], where square brackets refer to concentration; thus [H+] = hydrogen ion concentration. When measuring pH, [H+] is in units of moles of H+ per liter of solution. pH 7 is defined as neutral
When referring to soils, pH values lower that 6 are generally referred to as acidic (sometimes called sour) soils, while pH values greater than 6 are considered neutral or alkaline ( basic) soils. Most plant species of interest in forest farming perform best in the pH range from 5-7, but a few are acidophiles (acid loving) such as blueberries which prefer pH in the range of 4-5. Although H+ ion (pH) is not a nutrient per se, soil pH has important effects on availability of soil nutrients, as illustrated in the figure below. The effects of pH on availability of Fe and Ca in particular are of greatest consequence for Forest Farming. As pH increases, the availability of Fe declines. In sensitive species this causes interveinal chlorosis (yellowing) (e.g. pin oak, silver maple). Availability of Ca on the other hand increases as soil pH increases from acidic to alkaline.
In field agricultural systems, the pH of acidic soils are often increased by addition of lime [Ca(CO3)2] and the pH of alkaline soils is sometimes deliberately decreased (acidified) by addition of acidic organic matter such as peat moss, or elemental sulfur (soil bacterial convert it to sulfuric acid). Deliberate alteration of soil pH is rarely practical in forest farming, but the ambient soil pH is worth knowing since it can affect your choice of non- timber forest crops. If your site is topographically variable (hilly) be sure to test pH in multiple locations since it can vary considerably between bottomland and upland soils.
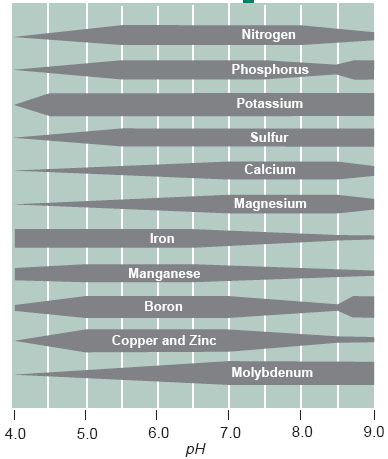
C. Jones and J. Jacobsen, 2001, Plant Nutrition and Soil Fertility, Montana State University Extension Service. Document can be downloaded from: http://www.montana.edu/wwwpb/pubs/mt4449.html.
Soil Organic Matter (OM)
OM is another important component of soil fertility and soil health. Although OM is not taken up directly and used by plants as a nutrient per se, it has important effects on soil fertility. Positively charged nutrient ions including the ammonium form of nitrogen (NH4+), potassium (K+), calcium (Ca+2), Magnesium (Mg+2) and others are attracted by the abundant negative electrical charges on the surface of OM particles (especially humus), and prevented from being leached away as water moves through the soil profile. Organic matter also has an important role to play in maintaining the structure and tilth of soils.
Soil Fertility Testing (Nutrient Analysis) Resources
New York
Pennsylvania
|
|

